DESIGN NAME:
i-STAT Alinity System
PRIMARY FUNCTION:
Point of Care Testing Device
INSPIRATION:
The source of inspiration for the product is patients who have to wait to get their blood results for a long time before their medical treatment can start. It is the premature baby waiting in a Neonatal Intensive Care Unit or the patient in an intensive care unit waiting for the blood result to come from the lab or an elderly person in a remote area with no access to a lab.
UNIQUE PROPERTIES / PROJECT DESCRIPTION:
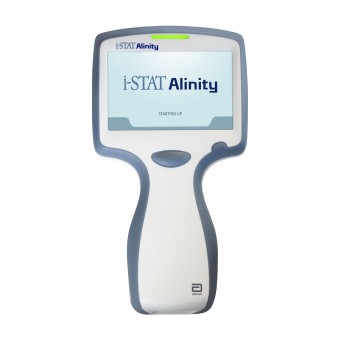
Abbott's i-STAT Alinity is a portable, handheld device that analyzes a broad range of blood tests and includes key features such as an ergonomic and durable design; a large, bright, color touch screen; lights and sound notifications and a high-resolution camera. It can be used in a variety of care settings such as hospitals, ambulances, urgent care, or remote areas. All materials are compliant to European Reduction of Hazardous Substances and European Waste Electrical and Electronic Equipment.
OPERATION / FLOW / INTERACTION:
The instrument is designed to be taken to the patient’s bedside or a convenient location. The instrument requires i‑STAT single-use cartridges containing electrodes and sensors to perform quantitative diagnostic testing on whole blood. After the insertion of a filled test cartridge, the instrument carefully monitors and controls the testing process. The only user intervention is in the form of data entry. Data entry is performed via the touchscreen or by barcode capture.
PROJECT DURATION AND LOCATION:
Project started in 2011 and received FDA clearance of the instrument and sodium assay on July 2016. The product was designed, developed and tested in Princeton, New Jersey, USA and Ottawa, Canada.
FITS BEST INTO CATEGORY:
Medical Devices and Medical Equipment Design
|
PRODUCTION / REALIZATION TECHNOLOGY:
The i‑STAT Alinity instrument includes the following subsystems:
Analytical measurement module: interfaces with the i‑STAT single-use test cartridges and controls execution of the measurement test cycle
User module: a central computing unit with embedded firmware that controls the user interaction with the device and supports communication with outside peripherals
User interface: allows data input, display of information, audio and visual alerts
Rechargeable battery
SPECIFICATIONS / TECHNICAL PROPERTIES:
255.8 mm (10.07 in) X 142.5 mm (5.61 in) X 81.2 mm (3.20 in)
660 g (1.45 lb) without battery; 840 g (1.85 lb) with battery
TAGS:
Point of care, POC, blood gas analyzer, blood analyzer, diagnostics, ivd, abbott point of care, APOC, i-STAT, i-STAT System
RESEARCH ABSTRACT:
Sodium, glucose and hematocrit assays (tests) were used to demonstrate the analytical functioning of the i-STAT Alinity instrument. Analytical evaluation of each test included multi-day precision, linearity, analyte recovery, limit of quantitation (LoQ) and interference from potential substances. The three method comparison studies used a total of 591 patient samples that were tested near patient (at the point of care) by the end user on both the i-STAT Alinity and i-STAT 1W instruments.
CHALLENGE:
The most difficult challenge in developing the i-STAT Alinity System is delivering lab-quality results typically expected of large instruments in a handheld device. Customers need the flexibility to bring the instrument with them to an Ambulance, a Clinic or from patient to patient in a hospital. Many customers carry the instrument in their hand or under their arms for easy transportation so they require a portable device that is small and lightweight, yet provides lab-quality results.
ADDED DATE:
2016-09-30 19:44:04
TEAM MEMBERS (1) :
IMAGE CREDITS:
Abbott
|