PulmoJet® Dry Powder Inhaler (DPI) by Sanofi Generics |
Home > Winners > #46609 |
 |
|
||||
| DESIGN DETAILS | |||||
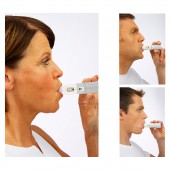
| DESIGN NAME: PulmoJet® PRIMARY FUNCTION: Dry Powder Inhaler (DPI) INSPIRATION: Correct inhaler use is essential for effective drug delivery but serious errors are common with current inhaler products, which may lead to poor disease control (Lavorini & Corbetta, 2008; Molimard, et al., 2003; Melani AS, et al., 2011). The respiratory market has increased opportunities for new and innovative products to provide better patient benefits and value (Lavorini & Corbetta, 2008; Thomas, et al, 2009; Lavorini, et al., 2008; Lavorini, 2013; Virchow, et al., 2008). UNIQUE PROPERTIES / PROJECT DESCRIPTION: PulmoJet® is an innovative, simple-to-use dry-powder inhaler with dose feedback mechanisms, giving physicians and patients confidence that the correct dose is delivered. It has a simple design for reliable and effective drug delivery. PulmoJet® is used by following 3 simple steps: open, inhale and close. The device includes audio and visual confirmation on correct dosing. OPERATION / FLOW / INTERACTION: PulmoJet® is an innovative, simple-to-use and easy-to-teach inhaler with multiple mechanisms, which is less prone to serious handling errors (Chrystyn et al., 2016). The device has three simple steps for effective use: open, inhale, close. The dose indicator shows you how many doses you have remaining. The green dose ready window will disappear after taking a dose. To load the next dose of medicine, the white cover is twisted back on to the inhaler until it clicks. PROJECT DURATION AND LOCATION: - FITS BEST INTO CATEGORY: Medical Devices and Medical Equipment Design |
PRODUCTION / REALIZATION TECHNOLOGY: Stainless steel, silicon, polypropylene, thermoplastic elastomer and other materials have been used in the construction of the PulmoJet® design. The breathe actuator is responsible for the protection and release of the loaded dose. The de-aggregator ensures the medication particles are the ideal size for inhalation. Medication is stored in the protected powder reservoir and the formulation is kept under pressure by a spring which fixes the position of the powder. SPECIFICATIONS / TECHNICAL PROPERTIES: 29.5 mm x 29.5 mm x 99 mm TAGS: DPI, simple-to-use, easy-to-teach, inhaler, open inhale close RESEARCH ABSTRACT: A Sanofi-led HI-TEC (Handling Inhalers – Technique Error Comparison) study assessed use and serious handling errors with PulmoJet® versus commonly used DPIs in asthma and COPD patients in a real-life setting. There was a significant risk reduction in serious handling errors reported from the randomized, crossover study. Patients using PulmoJet® made significantly fewer serious handling errors versus commonly used DPIs when trained using an instructional leaflet, and this benefit increases after additional video training (Chrystyn et al., 2016). CHALLENGE: The latest GINA and GOLD guidelines highlight the importance of inhaler technique in good disease control. Up to 54% of asthma and COPD patients using their DPI (dry powder inhaler) make serious handling errors (Al-Jahdali, et al., 2013). Failure to use inhalers correctly may potentially result in poor asthma and COPD control, increased costs and greater risk to the patient. Evidence has shown that reducing handling errors may lead to improved treatment adherence, and better disease and symptom control (Al-Jahdali, et al., 2013; Yildiz, et al., 2014; Small, et al., 2011; Voshaar, et al., 2014). PulmoJet® is an innovative DPI designed to reduce the serious handling errors frequently reported with the common devices used by asthma and COPD patients. ADDED DATE: 2016-02-26 11:46:28 TEAM MEMBERS (1) : IMAGE CREDITS: Sanofi Generics |
||||
| Visit the following page to learn more: http://www.sanofigenerics.com/ | |||||
| AWARD DETAILS | |
 |
Pulmojet® Dry Powder Inhaler (dpi) by Sanofi Generics is Winner in Medical Devices and Medical Equipment Design Category, 2015 - 2016.· Read the interview with designer Sanofi Generics for design PulmoJet® here.· Press Members: Login or Register to request an exclusive interview with Sanofi Generics. · Click here to register inorder to view the profile and other works by Sanofi Generics. |
| SOCIAL |
| + Add to Likes / Favorites | Send to My Email | Comment | Testimonials | View Press-Release | Press Kit |
Did you like Sanofi Generics' Medical Product Design?
You will most likely enjoy other award winning medical product design as well.
Click here to view more Award Winning Medical Product Design.